|
4/20/2023 0 Comments Chemical equation calculator  
In the previous step, we actually ended up balancing the oxygen atoms as well!īut sadly, this is still an unbalanced equation □. If we count up the number of atoms again, we have 4 H and 2 O on both sides, but there is 1 sodium atom on the left side, and 2 sodium atoms on the right side. We could have tried, but would have realized that we would need to balance H again (hence the trial and error). You might wonder why we didn't instead increase the number of H2 molecules. As a next step, lets balance H again by putting a 2 in front of NaOH so the equation reads: This is common and doesn't mean any mistakes were made.Īs was mentioned before, we knew we had to update multiple coefficients to balance the hydrogen atoms. We went from 1 unbalanced element to multiple. We now have 4 H, 2 O and 1 Na atom on the left, but 3 H, 1 O and 1 Na atom on the right. 
Notice how the 2 in front of H2O is "distributed" to both the H 2 and the O. Because 3 is not divisible by 2, it means coefficients of multiple compounds containing H will need to be changed. 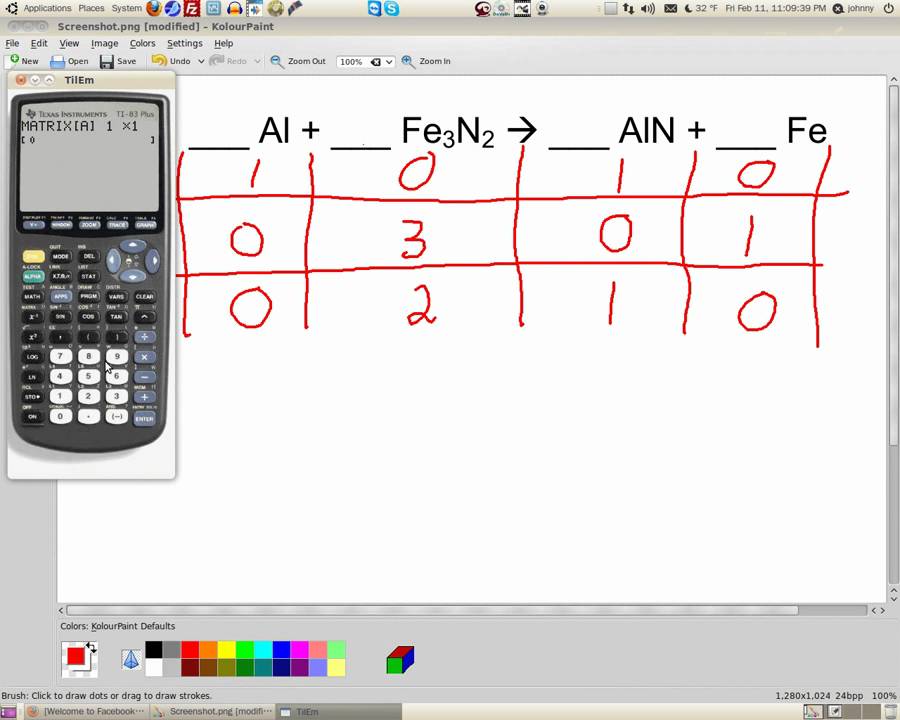
In this case, there are an equal number of Na and O atoms, but like last time, H needs to be balanced, with 2 on the left, and 3 on the right. Take for example the exothermic reaction of Sodium (Na) and Water (H2O), which releases heat, Sodium Hydroxide (NaOH) and Hydrogen Gas (H2): Usually, balancing chemical equations will require multiple steps. The previous example was simpler than most but demonstrated the basic concepts. That is the case in chemical equations like:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
